Regional Players Dominate the Regenerative Medicines Market, Despite the Presence of ~200 Competitors Comprising a Significant Number of Global and Country Niche Players, finds a recent market study on the Global Regenerative Medicines Market by Ken Research
Regenerative medicine is a process to restore human cells, tissues, and organs. It is referred to as a group of biomedical approaches to clinical therapies involving stem cells. Furthermore, therapies involving stem cells are, cell therapy, which is the injection of stem cells or progenitor cells, and tissue engineering, which is the transplantation of laboratory-grown organs and tissues.
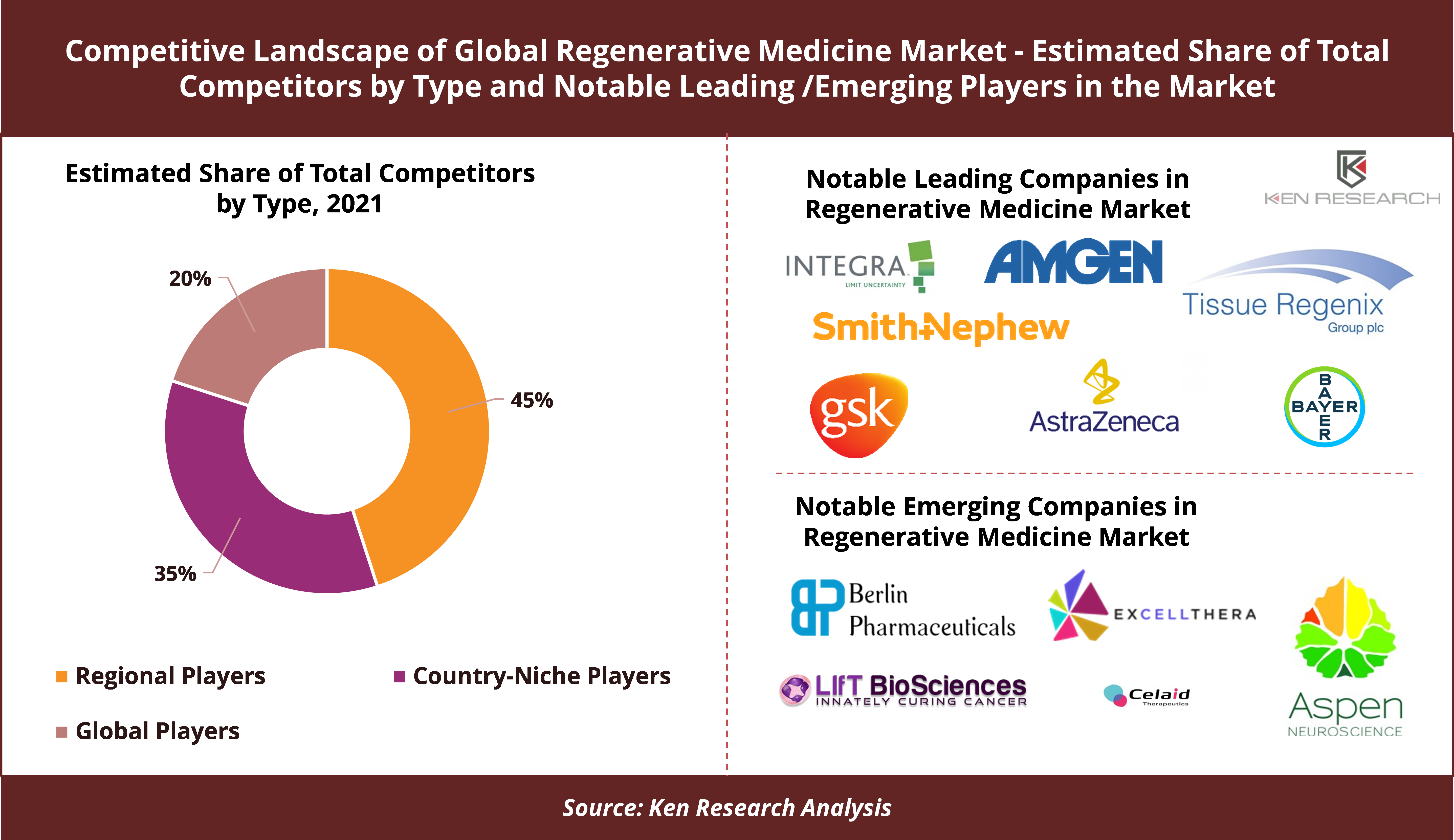
- Global Players Constitute ~20% of the Total Number of Competitors, While Regional Players Dominate Representing ~45% of Total Competitors
Competitive Analysis by the research study found that the global regenerative medicines market is highly competitive with ~200 players which include country-niche players, regional players as well as a significant number of globally diversified players, who deal in medical testing and surgeries for severe diseases, infections, and wounds. The large global players even though comprise about 20% in terms of the number of companies, hold a significant portion of the market revenue share followed by regional players which account for a major chunk of the market revenue. Most of the players are having their headquarters in the Europe region.
- Regular Product Improvisation and Internal Riveraly by the Majority of Players.
Detailed comparative analysis of key competitors available within the research study reveals that players have a variety of regenerative medicines products catering to various applications such as cancer, wound healing, diseases, trauma, disorder, brain injury, and others. All leading competitors have a strong presence in the global regenerative medicines market. Furthermore, several companies and organizations are aggressively investing in R&D to cater to the demand of end-users.
- In September 2022, the Centre for Breakthrough Medicines (CBM) and jCyte announced a multi-year manufacturing agreement wherein CBM will be the prime manufacturer of jCyte’s U.S. Food & Drug Administration’s Regenerative Medicine Advance Therapy (RMAT) designated cell therapy, jCell.
- In March 2021, Amgen Inc., a USA-based company, announced an agreement to acquire Rodeo Therapeutics Corporation, a privately held biopharmaceutical company based in Seattle that develops small-molecule therapies designed to promote regeneration and repair of multiple tissues.
Increased Adoption Of Regenerative Medicines Globally Leading To Major Industry-Wide Developments, Acquisitions, Collaborations
- In May 2020, the Global Institute of Stem Cell Therapy and Research (GIOSTAR), received FDA approval for Covid19 clinical trials with stem cells to treat the Covid19 patients.
- In May 2020, According to United Arab Emirates Ministry of Foreign Affairs & International Corporation, stated that a patent has been granted by the Ministry of Economics for the development of an innovative and promising treatment for Covid19 infection using stem cells.
For more information on the research report, refer to the below link:
Competitors in Regenerative Medicines Sector | Trends in the Regenerative Medicine Market
Read Also –
3 Key Insights on the US$ 70 Billion Opportunity in the Global Regenerative Medicine Market
Follow Us –
LinkedIn | Instagram | Facebook | Twitter | YouTube
Contact Us: –
Ken Research
Ankur Gupta, Head Marketing & Communications
+91-9015378249